Interim Infection Prevention and Control Recommends to Prevent SARS-CoV-2 Spread in Nursing Homes
November 22, 2021Summary of Recent Changes
- Updated outbreak response guidance to promote use of contact tracing approach. Alternative broad-based approaches to outbreak response at a facility-wide or unit level are also described.
- Updated expanded screening testing recommendations for healthcare personnel (HCP).
- Updated recommendations for quarantine of fully vaccinated residents.
- Updated visitation guidance.
Key Points
- Older adults living in congregate setting are at high risk of being affected by respiratory and other pathogens, such as SARS-CoV-2.
- A strong infection prevention and control (IPC) program is critical to protect both residents and healthcare personnel (HCP).
- Even as nursing homes resume normal practices, they must sustain core IPC practices and remain vigilant for SARS-CoV-2 infection among residents and HCP in order to prevent spread and protect residents and HCP from severe infections, hospitalizations, and death.
In general, healthcare facilities should continue to follow the IPC recommendations for unvaccinated individuals (e.g., use of Transmission-Based Precautions for those that have had close contact to someone with SARS-CoV-2 infection) when caring for fully vaccinated individuals with moderate to severe immunocompromise due to a medical condition or receipt of immunosuppressive medications or treatment.
Other factors, such as end-stage renal disease, likely pose a lower degree of immunocompromise and there might not be a need to follow the recommendations for those with moderate to severe immunocompromise. However, fully vaccinated people in this category should consider continuing to practice physical distancing and use of source control while in a healthcare facility.
Ultimately, the degree of immunocompromise for the patient is determined by the treating provider, and preventive actions are tailored to each individual and situation.
Infection Prevention and Control Program
Assign one or more individuals with training in infection control to provide on-site management of the IPC program.
- This should be a full-time role for at least one person in facilities that have more than 100 residents or that provide on-site ventilator or hemodialysis services. Smaller facilities should consider staffing the IPC program based on the resident population are facility service needs identified in the IPC risk assessment.
- CDC has created an online training course that can orient individuals to this role in nursing homes.
Provide supplies necessary to adhere to recommended IPC practices
- Ensure HCP have access to all necessary supplies including alcohol-based hand sanitizer with 60-95% alcohol, personal protective equipment (PPE), and supplies for cleaning and disinfection.
- Put FDA-approved alcohol-based hand sanitizer with 60-95% alcohol in every resident room (ideally both inside and outside of the room) and other resident care and common areas (e.g., outside dining hall, in therapy gym.)
Educate residents, HCP, and visitors about SARS-CoV-2, current precautions being taken in the facility, and actions they should take to protect themselves.
Vaccinations
Vaccinated residents and HCP against SARS-CoV-2
- Vaccination acceptance among nursing home personnel remains low, but is critical to protect both staff and residents against COVID-19. CDC continues to stress the importance of getting vaccinated when it is offered to you.
- Per CMS requirements, weekly vaccination numbers of nursing home residents and HCP must be reported into the NHSN LTCF Weekly HCP & Resident COVID-19 Vaccination Reporting module.
Source Control and Physical Distancing Measures
Refer to Interim Infection Control Recommendations for Healthcare Personnel During the COVID-19 Pandemic for details regarding source control and physical distancing measures recommended for vaccinated and unvaccinated HCP and residents.
Visitation
Have a plan for visitation
- Send letters or emails to families and post signs at entrances reminding them of the importance of getting vaccinated, recommendations for source control and physical distancing and any other facility instructions related to visitation, including not to visit if they have any of the following:
- A positive viral test for SARS-CoV-2,
- Symptoms of COVID-19, or
- If they currently meet criteria for quarantine
- Facilitate and encourage alternative methods for visitation (e.g., video conferencing) and communication with the resident.
Additional information about visitation for nursing homes and intermediate care facilities for individuals with intellectual disabilities and psychiatric residential treatment facilities is available from CMS.
Personal Protective Equipment
Ensure proper use, handling and implementation of personal protective equipment
- Information is available:
Testing
Create a plan for testing residents and HCP for SARS-CoV-2
- Anyone with even mild symptoms of COVID-19, regardless of vaccination status, should receive a viral test as soon as possible.
- Asymptomatic HCP with a higher-risk exposure and residents with close contact with someone with SARS-CoV-2 infection, regardless of vaccination status, should have a series of two viral tests for SARS-CoV-2 infection. In these situation, testing is recommended immediately (but not earlier than 2 days after exposure) and, if negative, again 5-7 days after the exposure. Criteria for use of post-exposure prophylaxis are described elsewhere.
Evaluating and Managing Personnel and Residents
Identify space in the facility that could be dedicated to monitor and care for residents with confirmed SARS-CoV-2 infection
- Determine the location of the COVID-19 care unit and create a staffing plan.
- The location of the COVID-19 care unit should ideally by physically separated from other rooms or units housing residents without confirmed SARS-CoV-2 infection. This could be a dedicated floor, unit, or wing in the facility or a group of rooms at the end of the unit that will be used to cohort residents with SARS-CoV-2 infection.
- Identify HCP who will be assigned to work only on the COVID-19 care unit when it is in use. At a minimum, this should include the primary nursing assistants (NAs) and nurses assigned to care for these residents. If possible, HCP should avoid working on both the COVID-19 care unit and other units during the same shift.
- To the extent possible, restrict access of ancillary personnel (e.g., dietary) to the unit.
- Ideally, environmental services (EVS) staff should be dedicated to this unit, but to the extent possible, EVS staff should avoid working on both the COVID-19 care unit and other units during the same shift.
- To the extent possible, HCP dedicated to the COVID-19 care unit (e.g., NA and nurses) will also be performing cleaning and disinfection of high-touch surfaces and shared equipment when in the room for resident care activities. HCP should bring an Environmental Protection Agency (EPA)- registered disinfectant (e.g., wipe) from List N into the room and wipe down high-touch surfaces (e.g., light switch, doorknob, bedside table) before leaving the room.
Manage Residents with Close Contact
Manage residents who had close contact with someone with SARS-CoV-2 infection
- Unvaccinated residents who have had close contact with someone with SARS-CoV-2 infection should be placed in quarantine for 14 days after their exposure, even if viral testing is negative. HCP caring for them should use full PPE (gowns, gloves, eye protection, and N95 or higher-level respirator).
- Although not preferred for healthcare settings, options for shortening quarantine are available.
- Fully vaccinated residents who have had close contact with someone with SARS-CoV-2 infection should wear source control and be tested as described in the testing section. Fully vaccinated residents and residents with SARS-CoV-2 infection in the last 90 days do not need to be quarantined, restricted to their room, or cared for by HCP using the full PPE recommended for the care of a resident with SARS-CoV-2 infection unless they develop symptoms of COVID-19, are diagnosed with SARS-CoV-2 infection, or the facility is directed to do so by the jurisdiction’s public health authority. Additional potential exceptions are described here.
Definitions:
Healthcare Personnel (HCP): HCP refers to all paid and unpaid persons serving in healthcare settings who have the potential for direct or indirect exposure to patients or infectious materials, including body substances (e.g., blood, tissue, and specific body fluids); contaminated medical supplies, devices, and equipment; contaminated environmental surfaces; or contaminated air. HCP include, but are not limited to, emergency medical service personnel, nurses, nursing assistants, home healthcare personnel, physicians, technicians, therapists, phlebotomists, pharmacists, dental healthcare personnel, students and trainees, contractual staff not employed by the healthcare facility, and persons not directly involved in patient care, but who could be exposed to infectious agents that can be transmitted in the healthcare setting (e.g., clerical, dietary, environmental services, laundry, security, engineering and facilities management, administrative, billing, and volunteer personnel).
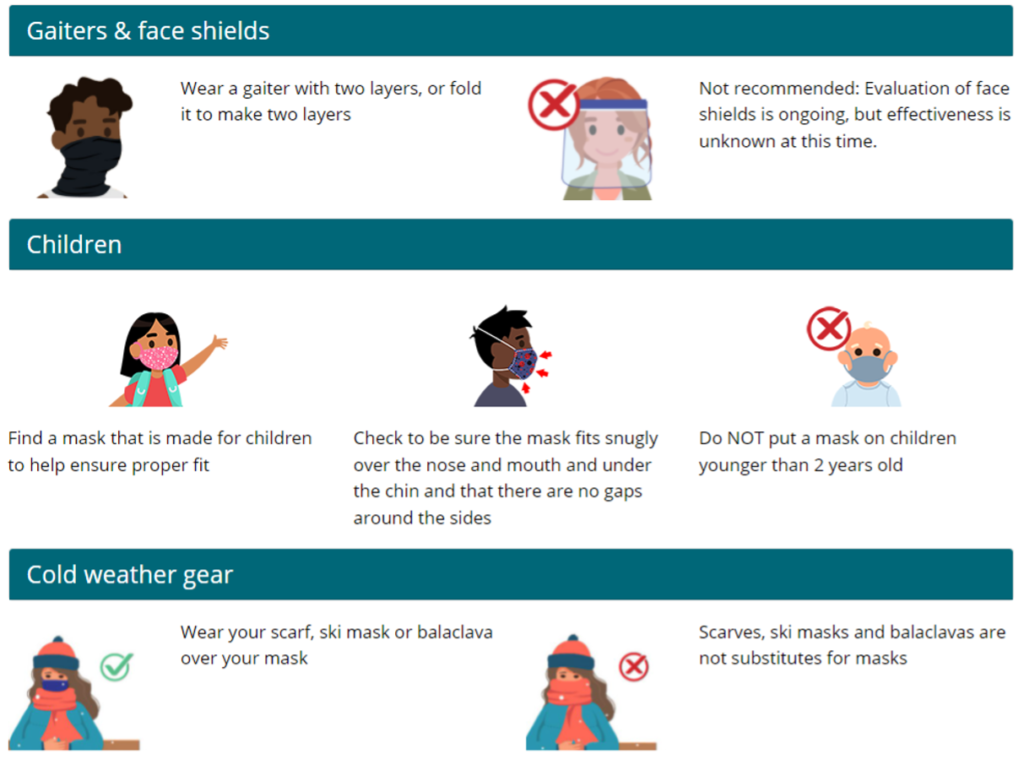
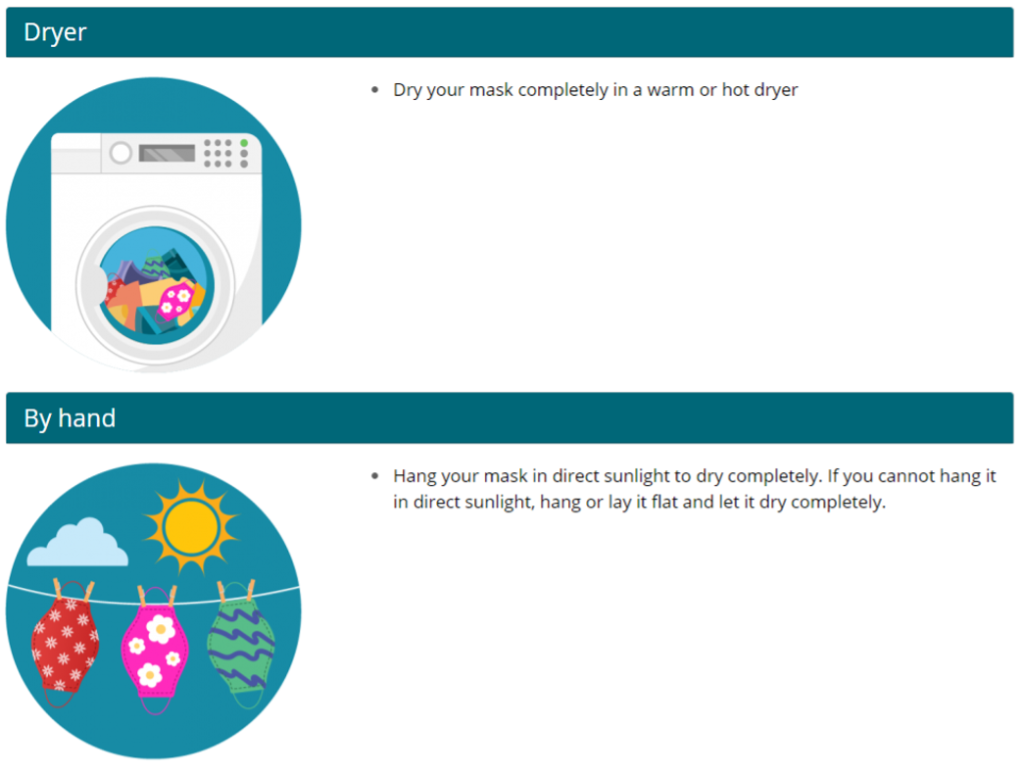
Source Control: Use of well-fitting cloth masks, facemasks, or respirators to cover a person’s mouth and nose to prevent spread of respiratory secretions when they are breathing, talking, sneezing, or coughing. Cloth masks, facemasks, and respirators should not be placed on children under the age of 2, anyone who cannot wear one safely, such as someone who has a disability or an underlying medical condition that precludes wearing a cloth masks, facemask, or respirator safely, or anyone who is unconscious, incapacitated, or otherwise unable to remove their cloth mask, facemask, or respirator without assistance. Face shields alone are not recommended for source control.
Respirator: A respirator is a personal protective device that is worn on the face, covers at least the nose and mouth, and is used to reduce the wearer’s risk of inhaling hazardous airborne particles (including dust particles and infectious agents), gases, or vapors. Respirators are certified by CDC/NIOSH, including those intended for use in healthcare.
Nursing Home-onset SARS-CoV-2 Infections: refers to SARS-CoV-2 infections that originated in the nursing home. It does not refer to the following:
- Residents who were known to have SARS-CoV-2 infection on admission to the facility and were placed into appropriate Transmission-Based Precautions to prevent transmission to others in the facility.
- Residents who were placed into Transmission-Based Precautions on admission and developed SARS-CoV-2 infection within 14 days after admission.
To learn more, please visit https://www.cdc.gov/coronavirus/2019-ncov/hcp/long-term-care.html